Technical terms of quality management: D
DAA
Department Activity Analysis.
DACH
Deutsche Akkreditierungsstelle Chemie GmbH, Frankfurt. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s National Accreditation Body as of January 1, 2010. The DACH entered the DAkkS via the DGA.
DAkkS
Deutsche Akkreditierungsstelle GmbH, Berlin. Germany‘s National Accreditation Body. The organization started its activities on January 1, 2010 and bundles all accreditation assignments in Germany. www.dakks.de
Danger
Situation in which the existing risk is greater than the acceptable risk. (DIN VDE 31000-2)
DAP
- Deming Application Prize.
- Deutsches Akkreditierungssystem für Prüfwesen GmbH, Berlin. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body as of January 1, 2010. The DAP entered the DAkkS via the DGA. www.dakks.de
DAR
Deutscher Akkreditierungsrat. The obligations for monitoring were moved to the DAkkS as of January 1, 2010.
DASET
Deutsche Akkreditierungsstelle Stahlbau und Energietechnik, Düsseldorf. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body as of January 1, 2010.
DASMIN
Deutsche Akkreditierungsstelle Mineralöl GmbH, Hamburg. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body as of January 1, 2010.
DATech
Deutsche Akkreditierungsstelle für Technik, Frankfurt. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body as of January 1, 2010.
DAU
Deutsche Akkreditierungs- und Zulassungsgesellschaft für Umweltgutachter mbH, Bonn. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body as of January 1, 2010.
Declaration of Conformity
Defect
Non-fulfillment of a requirement, so that an intended or specified use is not possible. Due to the legal meaning (particularly in association with product liability) ISO 9000:2015 points out the conceptual difference between defect and nonconformity and recommends using the term “defect“ with extreme caution. There can also be a defect when the use intended by the customer is impaired by the information provided (e.g., operating or maintenance instructions). (On the basis of ISO 9000:2015)
Delivery inspection
Acceptance inspection that is performed before a product is delivered. It is used to prove that the requirements or technical terms of delivery have meet fulfilled. This inspection can also be conducted at the same time as the final inspection. The contractor is responsible for the delivery inspection (as far as no agreements to the contrary have been reached). This can be either an internal or external contractor. (On the basis of DIN 55350-17)
Delivery lot
One or more lots that are delivered as a whole at one time.
(DGQ Volume 11-04:2009)
Delphi method
Systematic questioning procedure that comprises several steps. This method is used to allow a better assessment of trends, technical developments or, for example, future events.
Deming
The American William Edwards Deming (1900 –1993) was one of the thinkers and significant figures in quality science. His work focused primarily on the “14-point management program“ and the “principle of continual improvement.“
Deming cycle
Demonstration requirement
Requirement for demonstration of the implementation of management elements with respect to the customer or the client in the case of a contractual agreement, or with respect to a competent body in the case of legal requirement.
Note: Demonstration requirements result from the fact that the quality of complex products cannot be determined by an incoming inspection alone. (DGQ Volume 11-04:2009)
Dependability
Comprehensive term that describes availability and the factors that influence it: “reliability performance“, “maintainability performance“ and “maintenance support performance“. (On the basis of ISO 9000:2015)
Dependability requirement
Part of the quality requirement with respect to the behavior of a unit in terms of its dependability during or after stipulated time spans under stipulated use conditions. (On the basis of DIN 55350-11)
Design and development
Transforming processes that turn requirements into specified characteristics or into the specification of a product, process or system. (On the basis of ISO 9000:2015)
Design FMEA
See Failure Mode and Effects Analysis.
Design of experiments (DoE)
Those parts of experimental design in which a statistical procedure is used.
Remark 1: The Shainin method is the name for a strategy for problem-solving during ongoing production, especially with the help of statistical experimentation (DoE) methodology. The method involves determining the key problem areas by first using simple preliminary examinations, e.g., by means of comparing pairs. This is an attempt to reduce the number of influencing factors that are presumably responsible for the problem. Subsequently, the “k“ influencing factors remain. Their effects are examined in a “2k“ full factorial experimental design.
(DGQ Volume 11-04:2009)
Design review
Examination of a preliminary design from quality points of view. (On the basis of DIN 55350-17)
Design review technique
Design test
Systematic examination of a design and all partial outcomes. The objective is to check if the quality requirements are fulfilled. This test must be documented.
Desired value
Value of a quantitative characteristic. This characteristic‘s actual values should differ from the desired value as little as possible. (On the basis of DIN 55350-12)
Determination
Activity for identifying one or more characteristics (and their characteristic values). (On the basis of ISO 9000:2015)
Development model
(On the basis of DIN 55350-15)
Deviation
Difference between an observed value or characteristic value and a reference value or suitable mean value of the variable. (On the basis of DIN 55350-12)
Deviation permit
Permission to deviate from the requirements originally placed on a product. This permission is granted before the product realization. It is normally limited to a certain quantity of a product or for a certain time span and for a certain use. (On the basis of ISO 9000:2015).
DFA
Design for Assembly. Product design and development with ease of assembly considerations. (On the basis of IATF 16949:2016)
DFM
Design for Manufacturing. Product design and process planning with a view to easy, economical manufacturing. (On the basis of IATF 16949:2016)
DFMA
Design for Manufacturing and Assembly. Development and design in line with both DFM and DFA. (On the basis of IATF 16949:2016)
DFSS
= Design for Six Sigma
Six Sigma is applied as a tool both for existing processes and for processes that are to be created for the first time. The objective is to build up a process with Six Sigma capability ahead of time.
DGA
Deutsche Gesellschaft für Akkreditierung mbH, Frankfurt. The DGA was created on September 14, 2009 by the merger of the Deutsche Akkreditierungsstelle Chemie (DACH), the Trägergemeinschaft für Akkreditierung (TGA) and the Deutsches Akkreditierungssystem Prüfwesen. The organization became a part of the new Deutsche Akkreditierungsstelle DAkkS effective as of January 1, 2010.
DGQ
Deutsche Gesellschaft für Qualität e.V., Frankfurt. www.dgq.de
DGWK
Deutsche Gesellschaft für Warenkennzeichnung. DGWK was established in 1972 and today is called DINCERTCO Gesellschaft zur Konformitätsbewertung mbH, Berlin. www.dincertco.de
DGZfP
Deutsche Gesellschaft für Zerstörungsfreie Prüfung e.V., Berlin. www.dgzfp.de
DIN
Deutsches Institut für Normung e.V., Berlin. www.din.de
DIN SPEC
Technical specification that is published by the DIN.
Direct costs
Costs that can be attributed directly to the product.
DIS
Draft International Standard. Draft for a standard.
Discrete characteristic
Quantitative characteristic with a finite number or countably infinite number of forms. (On the basis of DIN 55350-12)
Discrete data
Discrimination threshold
Smallest change in an input value that can still be perceived by a measuring instrument.
Dispersion (also called variability)
Qualitative term for the deviation behavior of characteristic values.
Note: Some examples of quantitative statements on dispersion are variance, standard deviation and range. (DGQ Volume 11-04:2009)
Distribution
Statement on how how a characteristic is likely to behave. (On the basis of ISO 3534-1)
DITR
Deutsches Informationszentrum für Technische Regeln in the DIN. The DITR database contains bibliographical identification of around 300 sets of regulations.
DKD
Deutscher Kalibrierdienst, Braunschweig. The accreditation assignments were combined in a new structure in the DAkkS as Germany‘s national accreditation body.
DKE
Deutsche Kommission Elektrotechnik Elektronik Informationstechnik in the DIN and VDE, Frankfurt.
DMADV
Design – Measure – Analyze – Design – Verify
Project management method within Six Sigma which plays a role in the development of new processes.
DMAIC
Define – Measure – Analyze – Improve – Control: Project management methodology in Six Sigma projects (also known as the breakthrough strategy). Processes are structured, analyzed and optimized with the help of a project. The DMAIC methodology is systematically divided into the following five phases: Define (a problem), Measure (the influencing factors), Analyze (the influencing factors), Improve (the setting of the influencing factors), Control (or monitor the influencing factors).
Document
Information and the medium on which it is depicted (carrier medium). A document can be in paper form, electronic, a photograph, a master sample or a combination of these. ISO 9000:2015 lists as examples: Record, specification, procedure document, drawing, report and standard. Several documents (e.g., specifications and records) are also referred to as documentation. (On the basis of ISO 9000:2015)
Document control
Documented procedure for creating, checking, approving, identifying, distributing and updating documents. Document control also includes the identification and distribution of external documents, such as standards, customer specifications or customer drawings. If obsolete documents are stored, they must be identified in such an unambiguous manner that unintended use can be ruled out. Furthermore, not only documents from the management documentation should be subject to document control. Job descriptions, operating instructions, operating manuals for products, specifications, calculation bases or price lists that are used as specifications to fulfill customer requirements should also be taken into consideration. Records represent a special document type. Document control for records includes measures for the identification, storage, protection, retrieval, retention and disposition of records. (cf. ISO 9001:2015)
Documentation
All documents that belong to a system, project, process or product. See Document.
Documented information
Information (including its storage medium) which is subject to the control of documents. (On the basis of ISO 9000:2015)
DoE
Design of Experiments. See Statistical experimental design.
Dot plot
Graphics tool (also: one-dimensional dispersion diagram) used to show the acquired measured values on one axis. This is already sufficient to show the first data distributions. Each point is a measured value.
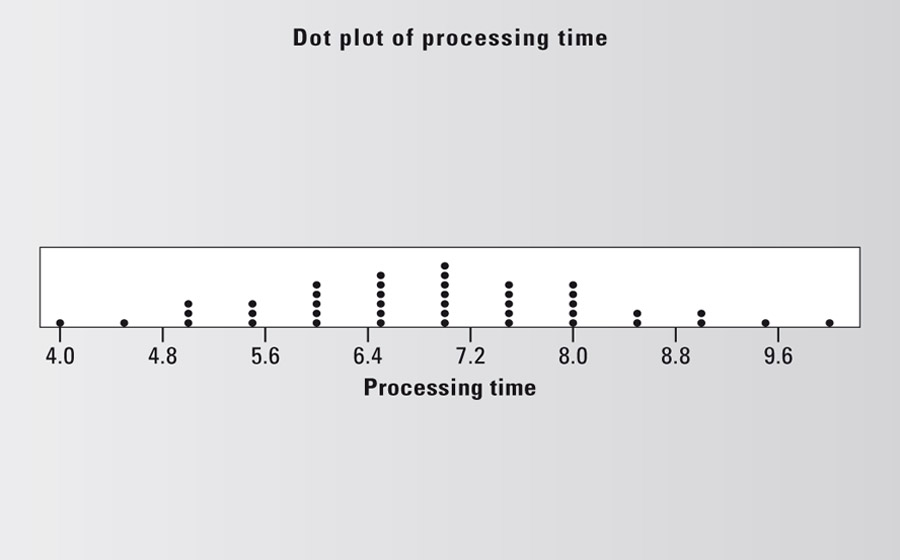
Downtime
Time during which a unit is not available due to an internal cause.
Note: Uptime and downtime together constitute the complete interval of the time between two consecutive failures for products that are to be maintained, with the uptime possibly containing breaks in operation. (DGQ Volume 11-04:2009)
DPMO
= Defects per Million Opportunities
Derived from the calculation of the DPO, but extrapolated to one million. The process indicator states how many defects would be made (on the basis of the produced units or sample) in one million opportunities for defects.
DPMO = DPO * 1.000.000
DPO
= Defects per Opportunity
Process indicator to measure the probabilities of defects in process steps. Here all the defects that occurred in the produced units or sample are set in relationship to all opportunities that the process/process step has to produce a defect.

DPU
= Defects per Unit
Process indicator to describe how many defects were produced per unit. The DPU takes the actual number of defects in relationship to the produced units or sample into consideration.
DPU = Sum of all defects / Sum of all produced units

